Mundo Nano | v16n31
http://dx.doi.org/10.22201/ceiich.24485691e.2023.31.69798 | Mundo Nano| julio-diciembre 2023
Review papers
Diana L. Mancera-Zapata*
Eden Morales Narváez**♦
*Centro de Investigaciones en Óptica, A. C. León, Guanajuato, México.
**Universidad Nacional Autónoma de México, Centro de Física Aplicada y Tecnología Avanzada, Biophotonic Nanosensors Laboratory.
Abstract:
Biosensing systems are powerful biotechnological tools which are widely applied in medical and environmental settings. Herein, we provide an overview of a recently developed optical biosensing system based on the quenching abilities of graphene oxide and fluorescent bioprobes. This biosensing platform has been demonstrated to be a fast, cost-effective and reliable nanophotonic technology. In particular, it has been exploited to detect relevant analytes in real matrixes, including prostate specific E. coli and COVID-19 antibodies. Besides, this technology enabled the detection of sialidase in clinical samples to determine bacterial vaginosis. This biosensing system has recently been used to determine relevant information on the kinetics of proteins involved in the biorecognition process, everything performed in real-time and in a single step.
Keywords: wash-free biosensing, nanophotonics, graphene-related materials, fluorescence quenching, protein detection
Resumen:
Los sistemas de biodetección son poderosas herramientas biotecnológicas que se aplican ampliamente en entornos médicos y ambientales. En este artículo, proporcionamos una descripción general de un sistema de biodetección óptica desarrollado recientemente, basado en las capacidades de extinción del óxido de grafeno y las biosondas fluorescentes. Se ha demostrado que esta plataforma de biodetección es una tecnología nanofotónica rápida, rentable y fiable. En particular, se ha aprovechado para detectar analitos relevantes en matrices reales, incluidos los anticuerpos contra E. coli y COVID-19 específicos de la próstata. Además, esta tecnología permitió la detección de sialidasa en muestras clínicas, para determinar la vaginosis bacteriana. Por último, este sistema de biodetección se ha utilizado recientemente para determinar información relevante sobre la cinética de las proteínas involucradas en el proceso de biorreconocimiento, todo realizado en tiempo real y en un solo paso.
Palabras clave: biodetección sin lavado, nanofotónica, materiales relacionados con el grafeno, extinción de fluorescencia, detección de proteínas
Fecha de aceptación: May 4 2023
Fecha de publicación: June 1 2023
Introduction
Biosensing systems are now a fundamental tool in healthcare, environmental monitoring, safety and protection. Nowadays, due to the pandemic outbreak generated by the SARS-CoV-2 virus, biosensors demonstrated a fundamental role offering reliable and low-cost results within a short response time to tackle such a pandemic scenario by means of timely diagnosis technologies (Huang et al., 2017).
Biosensors are analytical tools that facilitate the detection of (bio)molecules with a high degree of specificity and sensitivity. The fundamental idea behind biosensors consists of three main blocks (Figure 1), the first block consists of a biological recognition event or biorecognition event, which is extremely specific. The next block is the transduction and involves converting a biorecognition event into a measurable signal to acquire information related to the modulation of a specific physical phenomenon, such as fluorescence intensity, color change, etc. The resulting signal is then obtained and decoded (Srivastava et al., 2018; Velasco-Soto et al., 2015). It is important to note that biosensors with high sensitivity have the ability to produce a signal from smallest changes in analyte concentration. The limit of detection (LOD) of a biosensor, which is an indicator of analytical sensitivity, is determined by the smallest amount of analyte that the biosensor is able to detect (Vessman et al., 2001).
Figura 1. Schematic representation of different blocks integrating a biosensor.
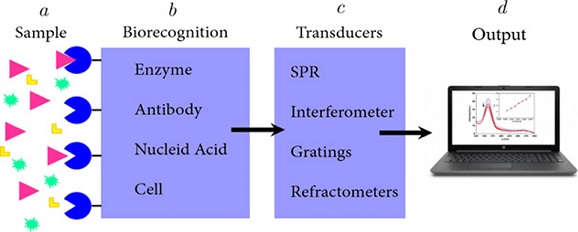
a) Sample. Substance to be analyzed in which is possible to detect the presence the target analyte. b) Biorecognition. This stage requires elements with high affinity against the target analyte. c) Transducers. A device that converts the measured biological activity into an analytical signal. d) Output. Measurable signal indicating the presence of the analyte in the sample.
Source: Taken from Diana L. Mancera (2018).
Herein, we provide an overview on a nanophotonic biosensing technology developed by our team. The biosensing platform is based on fluorescence or Förster resonance energy transfer (FRET) that takes place between fluorescent bioprobes acting as donors and graphene oxide (GO), acting as an acceptor. Unlike enzyme-linked immunosorbent assay (ELISA) in which blocking, separation and washing steps must be performed, this new technology allows an immunoassay to be realized through the use of a single antibody. The response of this new biosensing system can be interrogated in real-time and is observable within a few minutes (e.g., 5 minutes). Additionally, this system works with diverse photoluminescent agents and analytes.
FRET-GO based biosensor
The biosensing mechanism
The operational mechanism of our FRET-GO based biosensing platform is based on fluorescence quenching generated by the non-radiative phenomenon known as FRET (Figure 2A-B). In this non- radiative phenomenon, two materials are involved, a donor and an acceptor. FRET takes place when the emission spectrum of the donor overlaps the absorption spectrum of the acceptor. In addition, there must be certain distance between these molecules, particularly approximately 20 nm using graphene-related materials (Morales-Narváez y Merkoçi, 2018).
Figura 2. FRET mechanism. Schematic representation.
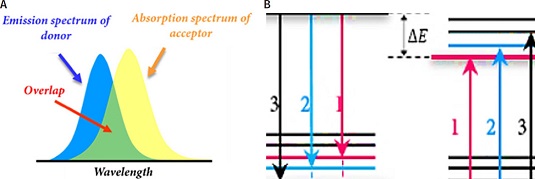
A. Overlapping between acceptor absorption spectrum and donor emission spectrum. B. Donor and acceptor molecular energy level schemes demonstrating correlated transitions. The vibrational transitions that occur in the donor molecule display the same energy as those that occur in the acceptor molecule.
Source: Taken from Diana L. Mancera (2018).
GO and its role in the biosensing platform system
Graphene is a carbon material. Due to its atomic thickness, it is considered a 2D material. It can be described as a planar sheet with several carbon atoms organized in a honeycomb arrangement which is the primary structure to build the others carbon allotropes (Figure 3) (Vasilios et al., 2015; Geim, Novoselov, 2007).
Figura 3. Representation of the structure of GO.
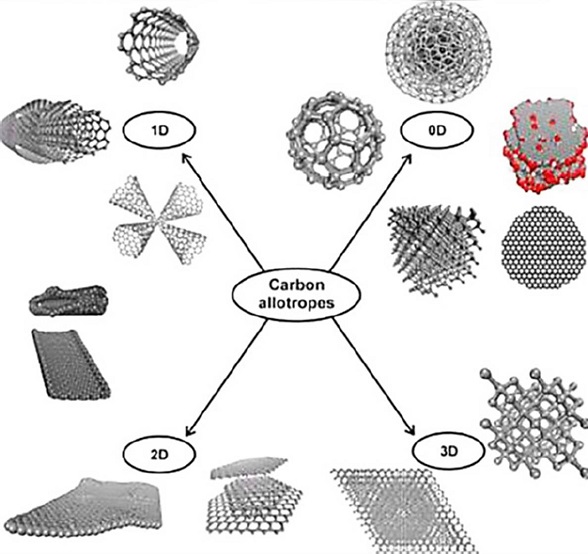
Source: Taken with permission from Vasilios et al. (2015). Copyright © 2015 American Chemical Society
Besides, the oxidized version of graphene, graphene oxide (GO), possesses functional groups such as hydroxyl, epoxy, carbonyl, and carboxyl groups. Particularly, GO exhibits carboxyl groups onto the edges and the other oxygen-containing groups on the basal plane, as Figure 4 shows (Morales-Narváez y Merkoçi, 2012).
Figura 4. Allotropes of carbon.
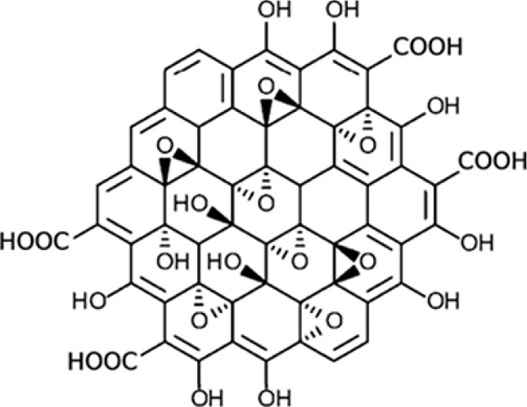
Source: Adapted with permission from Sinclair y Coveney (2019). Copyright © 2019 American Chemical Society
The Hummer approach is the most frequent method to produce GO. Importantly, ionic groups like O− and COO− found in GO enable this substance to engage electrostatically with proteins or DNA.
Literature reports the extraordinary optical properties of GO. For example, the wide spectral absorption range of GO is one of them. Hence, GO can be exploited as a universal acceptor in FRET (Forster, 1946). The proposed biosensing platform is featured by a surface coated with GO (FRET acceptor) and a fluorescent bioprobe (FRET donor). We discovered that the GO-coated surface has a strong affinity against those bioprobes that do not interact with the analyte, thus the respective fluorescence is quenched when no analyte is present in the sample; whereas, those fluorescent bioprobes interacting with the analyte are not quenched since such a complex (bioprobes-analyte) have no affinity against the GO-coated surface and the analyte also represents a spacer between the GO-coated surface and the bioprobes (Fluorophore-labelled antibody), as Figure 5 shows.
Figura 5. Operational mechanism of the biosensing plataform.
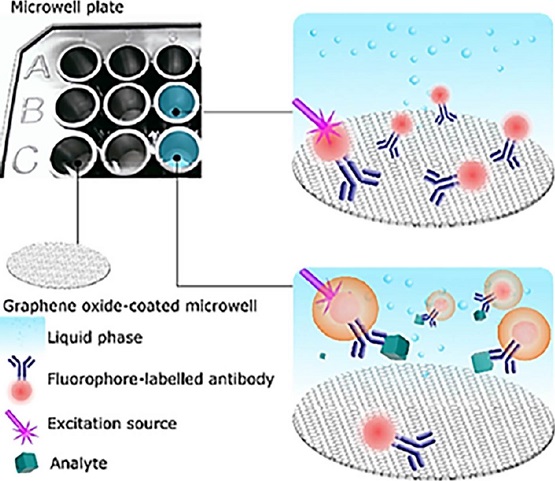
The GO-coated surface has a strong affinity against those bioprobes that do not interact with the analyte, thus the respective fluorescence is quenched when no analyte is present in the sample; whereas, those fluorescent bioprobes interacting with the analyte are not quenched since such a complex (bioprobes-analyte) have no affinity against the GO-coated surface and the analyte also represents a spacer between the GO-coated surface and the bioprobes.
Source: Reproduced with permission form Ortiz-Riaño et al. (2020). © 2020 Elsevier B.V. All rights reserved
FRET-GO based biosensor implementations
The proposed technology has been demonstrated to be a techically sound immunosensing platform. In general, microwell plates have been coated with GO to implement this novel technology (Ortiz-Riaño et al., 2020).
Costar 3603 are the microwell plates employed.1 The microwells surface is characterized to be hydrophilic, negatively charged, and consisting of 9-17% oxygen atoms (Li, 2008). Hence, the surface of these microwells is able to attach GO via hydrophilic contacts and hydrogen bonding given the aforementioned oxygen-containing groups of GO.
As a proof of concept, the FITC-labeled anti-human IgG antibodies (IgGFITC) with different concentrations of human IgG (H-IgG) were combined into GO-coated microwells (GOμW) and their photoluminescent response was explored in real-time. Figure 6A displays the optimal performance of this novel immunoassay targeting H-IgG. This analysis indicates that this immunodetection platform effectively reports the presence of the analyte by showing that the fluorescence levels (If/I0) are proportional to the concentration of the analyte. In addition, to evaluate the performance of this biosensing platform in a real matrix, human urine samples doped with different concentrations of prostate-specific antigen (PSA) were analyzed within 10 minutes, see Figure 6B-C.
Figura 6. Analytical performance of the immunosensing platform targeting HIgG and PSA.
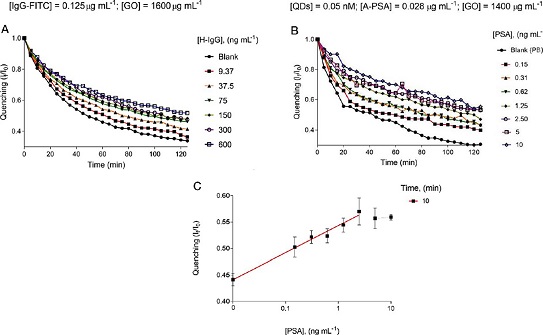
A. Real-time analysis of different concentrations of H-IgG and a blank sample. B. Real-time analysis of different concentrations of PSA and a blank sample. C. Calibration curve resulting at 10 minutes of the proposed immunoassay (PSA detection).
Source: Adapted with permission from Ortiz-Riaño et al. (2020). © 2020 Elsevier B.V. All rights reserved
In addition, this immunodetection platform was reported to exhibit an analytical sensitivity that is comparable to that of ELISA, with acceptable precision and accuracy (Table 1). Besides, other relevant advantages of this innovative immunodetection platform is that it can be interrogated in real-time, works with a single antibody and avoids washing, blocking and separation steps.
Tabla 1 Comparison of this approach with ELISA in terms of assay time and LOD.
*IgG (Total) Human Uncoated ELISA Kit - Invitrogen. https://www.thermofisher.com/mx/es/home/life-science/antibodies/immunoassays/elisa-kits.html?gclid=EAIaIQobChMI_t-zqdeO_wIVhQ99Ch0vsAKvEAAYASAAEgJG2PD_BwE&ef_id=EAIaIQobChMI_tzqdeO_wIVhQ99Ch0vsAKvEAAYASAAEgJG2PD_BwE:G:s&s_kwcid=AL!3652!3!415798135406!p!!g!!invitrogen%20elisa!9205681679!93387333557&cid=bid_pca_iel_r01_co_cp1359_pjt0000_bid00000_0se_gaw_bt_pur_con.. **E. coli HCP ELISA Kit, 2G (F1020). https://www.abcam.com/products/elisa/e-coli-hcp-elisa-kit-hostcell-protein-ab240997.html. Source: Author’s elaboration. Taking advantage of the versatility of this novel GOμWs-based technology, a nanophotonic
immunoassay was developed for the diagnosis of bacterial vaginosis (BV), which is
an infectious vaginal disease caused by the overgrowth of bacteria naturally found
in the vagina, which disrupts its natural balance (Wu et al., 2019). The evaluation of the Amsel criteria (Amsel et al., 1983) and the Nugent score (Nugent et al., 1991) are typically used to make a clinical diagnosis of BV. Both approaches rely on observations
made by highly qualified professionals who have even undergone extensive training.
In BV infection, the enzyme known as sialidase (SLD) plays a very important role since
several studies have found a close relationship between its production and the development
of BV infection. Hence, SLD has been introduced as a potential biomarker of BV infection
(Van de Munckhof et al., 2019).
In this context, a one-step quantitative biodetection system was designed to detect
SLD concentrations. For this purpose, a monoclonal antibody (mAb) against SLD, developed
by our colleagues in Universidad Autónoma de Guerrero, was exploited. Several immunological
methods have shown that this mAb can recognize sialidase with high sensitivity (Cortés-Sarabia et al., 2020). Hence, in our immunoassay, mAb-decorated quantum dots (mAb-QD) were employed as
a fluorescent bioprobe to interrogate the presence of SLD and GOμWs were also employed.
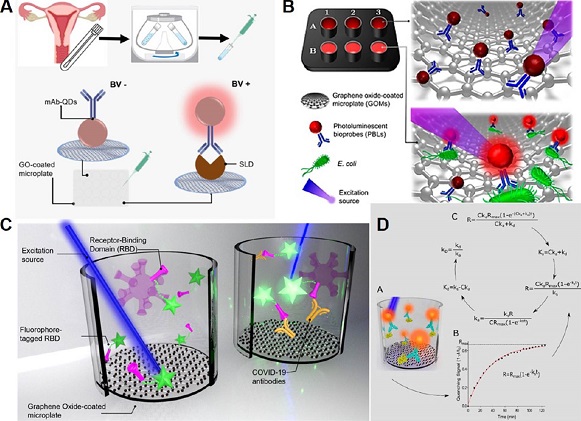
A schematic representation of our nanophotonic immunoassay targeting SLD is shown
in Figure 7A. With this nanophotonic immunoassay, 162 clinical samples were analyzed with a clinical
sensitivity/ specificity accounting for c.a. 96%. Besides, it was demonstrated as
a simple and cost-effective tool to detect BV, especially when compared with conventional
techniques such as indirect ELISA (Rodríguez-Nava et al., 2021).
Figura 7. Graphical representation of real-time biosensing platform in different approach and
analytes detection.
A. SLD Immunoassay diagram. B. Scheme of the biosensing platform targeting E. coli.
C. Biosensing system for COVID-19 seroconversion. D. Schematic representation of the
measurement of binding kinetics constants. Source: A: Adapted with permission from
Rodríguez-Nava et al. (2021).. B: Adapted with permission from Avila-Huerta et al. (2020),
Copyright © 2020 American Chemical Society. C: Adapted with permission from
Avila-Huerta et al. (2021).
Copyright © 2021, American Chemical Society. D: Adapted with permission from
Edwin et al. (2022).
Copyright © 2022, American Chemical Society. Food safety is another field in which having an efficient, fast, economical and at
the same time reliable biosensing system is of greatest importance since late detection
of pathogenic bacteria can be a great threat to human health as well as to the ecosystem.
Exploiting the same nanophotoic technology, we reported a rapid pathogen detection
platform (Avila-Huerta et al., 2020). Particularly, our biosensing platform was used to detect Escherichia coli, even
in cauliflower extracts, see Figure 7B.
The aforementioned configuration provided an optimum analytical performance at 30
minutes. As a result, the developed bacterial detection platform was shown to be quick
and extremely sensitive, with a limit of detection of around 2 CFU mL-1.
Our nanophotonic immunoassay was also adapted to offer a useful tool in the recent
pandemic scenario. In fact, we engineered an immunoassay targeting COVID-19 antibodies.
Particularly, based on the fact that the fluorophore-tagged recombinant SARS-CoV-2
spike receptor binding domain protein (F-RBD) can act as a bioprobe that emits fluorescence
upon detection of the COVID-19 antibody, a biosensing system for determination of
COVID-19 seroconversion was developed (Figure 7C). The cost of this test is about 0.5 USD per unit (laboratory scale). In addition,
the biosensing system provides optimal results within 42 min. With the intention of
proving that this technology is technically sound in a relevant environment, 34 human
serum samples were examined, as well (Avila-Huerta et al., 2021).
So far, our biosensing platform has been discussed as a versatile, low-cost, efficient
and advantageous tool, especially when compared with conventional techniques such
as ELISA. However, the most recent work in which this system is involved concerns
the study of the macromolecular association between the biorecognition probe and the
corresponding analyte. It is known that macromolecular association is crucial in drug
development, diagnostics and gene editing, however macromolecular association study
requires expensive instrumentation and complicated procedures (Patel et al., 2017). Since the developed nanophotonic immunoassay operates in real-time, binding constants
displayed during protein-protein association can also be determined by means of our
innovative technology. Figure 7D shows a schematic representation of this innovative technology to determine protein
kinetics. This one-step platform was used to determine the binding data for three
protein pairings, and the results were compared to data provided by suppliers or the
literature, indicating that this approach is comparable with existing methods. These
pairs consist of: 1) human immunoglobulin G (H-IgG)-fluorophore-tagged anti-H-IgG;
2) prostate-specific antigen (PSA)-quantum dot-tagged anti-PSA, and, 3) recombinant
anti-RBD protein-fluorophore-tagged SARS-CoV-2 spike receptor-binding domain. Additionally,
an open-source software that determines protein binding kinetic constants was offered
(Edwin et al., 2022).
We discussed that graphene oxide-coated microwells, GOμWs, combined with photoluminescent
bioprobes are a powerful tool for the implementation of an advantageous biosensing
system, which can be considered a highly transformative immunoassay in a standard
format (the 96-microwell format). Moreover, it is important to note that, due to the
universal fluorescence quenching ability of GO, it is possible to engineer a photoluminescent
immunodetection platform operating at different wavelengths, which may be useful for
future multiplexed applications. Besides, the proposed nanophotonic immunoassay can
be transferred to on-site applications using miniaturized technologies involving microfluidics
and/or paper-based analytical devices. In addition, to improve the clinical sensitivity
and specificity of our biosensing system, computational tools such as artificial intelligence
can be employed.
Amsel, R., Totten, P. A., Spiegel, C. A., Chen, K. C., Eschenbach, vD., and Holmes,
K. K. (1983). Nonspecific vaginitis. Diagnostic Criteria and Microbial and Epidemiologic
Associations. Am. J. Med., 74(1): 14-22. Avila-Huerta M. D, Ortiz-Riaño, E. J., Mancera-Zapata, D. L., Cortés-Sarabia, K.,
and Morales-Narváez, Eden. (2021). Facile determination of Covid-19 seroconversion
via nonradiative energy transfer. ACS Sens. https://doi.org/10.1021/acssensors.1c00795. Avila-Huerta, M. D., Ortiz-Riaño, E. J., Mancera-Zapata, D. L., Morales-Narváez, E.
(2020). Real-time photoluminescent biosensing based on graphene oxidecoated microplates:
a rapid pathogen detection platform. Anal. Chem. 2020. https://doi.org/10.1021/acs.analchem.0c02200. Cortés-Sarabia, K., Rodríguez-Nava, C., Medina-Flores, Y., Marta-Ruíz, O., LópezMeza,
J. E., Gómez-Cervantes, M. D., Parra- Rojas, I., Illades-Aguiar, B., FloresAlfaro,
E., and Vences-Velázquez, A. (2020). Production and characterization of a monoclonal
antibody against the Sialidase of Gardnerella Vaginalis using a synthetic peptide
in a MAP8 format. Appl. Microbiol. Biotechnol., 104: 6173-6183. Forster, Th. (1946). Energiewanderung und Fluoreszenz. Naturwissenschaften, 33(6):
166-175. https://doi.org/10.1007/BF00585226. Geim, A. K., Novoselov, K. S. (2007). The rise of graphene. Nature Materials, 6(3): 183-191. https://doi.org/10.1038/nmat1849.
Huang, A., Li, W., Shi, S., Yao, T. (2017). Quantitative fluorescence quenching on
antibody-conjugated graphene oxide as a platform for protein sensing. Scientific Reports,
7(1): 40772. https://doi.org/10.1038/srep40772.
Li, D., Müller, M. B., Gilje, S., Kaner, R. B., Wallace, G. G. (2008). Processable
aqueous dispersions of graphene nanosheets. Nat. Nanotechnol., 3: 101. https://doi.org/10.1038/nnano.2007.451
Mancera-Zapata, Diana L. (2018). Reduction of graphene oxide and its impact on the
performance of a biosensing system. Unpublished Master’s thesis. Centro de Investigaciones
en Óptica, A.C. CIO Repositorio. http://cio.repositorioinstitucional.mx/jspui/handle/1002/1187.
Morales-Narváez, E., A. Merkoçi. (2012). Graphene oxide as an optical biosensing platform.
Advanced materials, 24: 3298. [Front cover article, highly cited paper]. https://doi.org/10.1002/adma.201200373.
Morales-Narváez, E., Merkoçi, A. (2018). Graphene oxide as an optical biosensing platform:
progress report. Adv. Mater., 31(6): 1805043. https://doi.org/10.1002/adma.201805043.
Najeeb, M. A., Ahmad, Z., Shakoor, R. A., Mohamed, A. M. A., Kahraman, R. (2017).
Talanta, 168: 52-61. https://doi.org/10.1016/j.talanta.2017.03.022.
Nugent, R. P., Krohn, M. A., and Hillier, S. L. (1991). Reliability of diagnosing
bacterial vaginosis is improved by a standardized method of Gram Stain interpretation.
J. Clin. Microbiol., 29 (2): 297-301. Ortiz-Riaño, E. J., Avila-Huerta, M. D., Mancera-Zapata, D. L., Morales-Narváez, E.
(2020). Microwell plates coated with graphene oxide enable advantageous real-time
immunosensing platform. Biosens. Bioelectron., 165: 112319. https://doi.org/10.1016/j.bios.2020.112319. Ortiz-Riaño, Edwin J., Diana L. Mancera-Zapata, Martha Ulloa-Ramírez, Fernando Arce-Vega,
and Eden Morales-Narváez. (2022). Measurement of protein kinetics using a liquid phase-based
biosensing platform. Analytical Chemistry, Article ASAP. https://doi.org/10.1021/acs.analchem.2c03305. Patel, K., Halevi, S., Melman, P., Schwartz, J., Cai, S., Singh, B. R. (2017). Biosensors,
7(3): 32. https://doi.org/10.3390/bios7030032. Rodríguez-Nava, C., Cortés-Sarabia, K., Avila-Huerta, M. D., Ortiz-Riaño, E. J., Estrada-Moreno,
A. K., Alarcón-Romero, L. C., Mata-Ruiz, O., Medina-Flores, Y., Vences-Velazquez,
A., Morales-Narváez. (2021). Nanophotonic sialidase immunoassay for bacterial vaginosis
diagnosis. ACS Pharmacol. Transl. Sci. https://doi.org/10.1021/acsptsci.0c00211. Sinclair, Robert C., and Peter V. Coveney. (2019). Modeling nanostructure in graphene
oxide: inhomogeneity and the percolation threshold. J. Chem. Inf. Model, 59, 6, 2741-2745.
https://doi.org/10.1021/acs.jcim.9b00114. Srivastava, S., Senguttuvan, T. D., Gupta, B. K. (2018). Highly efficient fluorescence
quenching with chemically exfoliated reduced graphene oxide. Journal of Vacuum Science
& Technology B., 36(4): 04G104. https://doi.org/10.1116/1.5026170. Steiner, D. J., Cognetti, J. S., Luta, E. P., Klose, A. M., Bucukovski, J., Bryan,
M. R., Schmuke, J. J., Nguyen-Contant, P., Sangster, M. Y., Topham, D. J., Miller,
B. L. (2020). Array-based analysis of SARS-CoV-2, other coronaviruses, and influenza
antibodies in convalescent COVID-19 patients. Biosens. Bioelectron, 169: 112643. https://doi.org/10.1016/j.bios.2020.112643. Van den Munckhof, E. H. A., van Sitter, R. L., Boers, K. E., Lamont, R. F., Te Witt,
R., le Cessie, S., Knetsch, C. W., van Doorn, L.-J., Quint, W. G. V., Molijn, A.,
and Leverstein-Van Hall, M. A. (2019). Comparison of amsel criteria, nugent score,
culture and two CE-IVD marked quantitative real-time PCRs with microbiota analysis
for the diagnosis of bacterial vaginosis. Eur. J. Clin. Microbiol. Infect. Dis. ,
38(5): 959-966. Vasilios Georgakilas, Jason A., Perman, Jiri Tucek, and Radek Zboril, (2015). Broad
family of carbon nanoallotropes: classification, chemistry, and applications of fullerenes,
carbon dots, nanotubes, graphene, nanodiamonds, and combined superstructures. Chem.
Rev., 115(11): 4744-4822. https://doi.org/10.1021/cr500304f. Velasco-Soto, M. A., Pérez-García, S. A., Alvarez-Quintana, J., Cao, Y., Nyborg, L.,
Licea-Jiménez, L. (2015). Selective band gap manipulation of graphene oxide by its
reduction with mild reagents. Carbon, 93: 967-973. https://doi.org/10.1016/j.carbon.2015.06.013. Vessman, J., Stefan, R. I., Staden, J. F. V., Danzer, K., Lindner, W., Burns, D. T.,
Fajgelj, A., Müller, H. (2001). Selectivity in analytical chemistry: (IUPAC Recommendations
2001). https://doi.org/10.1515/iupac.73.0808. Wu, S., Lin, X., Hui, K. M., Yang, S., Wu, X., Tan, Y., Li, M., Qin, A.-Q., Wang,
Q., Zhao, Q., Ding, P., Shi, K., and Li, X. J. (2019). A biochemiluminescent sialidase
assay for diagnosis of bacterial vaginosis. Sci. Rep., 9, 20024.
♦ Correspondence author: eden@fata.unam.mx
Analyte
Method
Assay time (min)
LOD (ng mL-1)
Reference
H-IgG
ELISA
360
1.60
(“IgG (Total) Human Uncoated ELISA Kit - Invitrogen”) *
GOµW
45
2.56
Ortiz-Riaño et al. (2020)
PSA
ELISA
360
0.10
Najeeb et al. (2017)
GOµW
90
0.02
Ortiz-Riaño (2020)
E. Coli
ELISA
180
3
(E. coli HCP ELISA Kit, 2G (F1020) **
GOµW
30
2 CFU mL-1
Avila-Huerta et al. (2020)
SLD
ELISA
360
0.031
Cortés-Sarabia et al. (2020)
GOµW
120
0.012
Rodríguez-Nava et al. (2020)
COVID-19 Antibodies
ELISA
(25-43)
pg mL-1 to ng mL-1
Steiner et al. (2020)
GOµW
42
0.003
Avila-Huerta et al. (2021)
Conclusions and outlook
Referencias
Notes